
Therapy Clinic Business Model: Why 2026 Is the Year Entrepreneurs Are Moving on This Opportunity
Therapy Clinic Business Model: Why 2026 Is the Year Entrepreneurs Are Moving on This Opportunity
A Market Analysis and Business Guide for Entrepreneurs Evaluating the Peptide Clinic Space

The Market Signal That Most Entrepreneurs Are Still Catching Up To
Between April 2025 and April 2026, Google searches for peptide therapy grew 281 percent. On TikTok the growth was 459 percent. On Instagram it was 412 percent. Searches for the specific cost of peptide therapy are up 300 percent in the same period. These are not the numbers of a niche category finding its early adopters. These are the numbers of a mainstream consumer market arriving at scale — and arriving faster than most entrepreneurs have adjusted their thinking to account for.
The peptide therapy clinic is now one of the most commercially compelling business opportunities in the cash-pay health space. Consumer demand is documented and accelerating. The regulatory environment is actively improving for entrepreneurs who structure their clinics correctly. The competitive landscape in most U.S. markets has not yet caught up to the scale of patient demand. And the financial model — built around compounded protocol memberships, recurring lab revenue, and cash-pay economics — produces the kind of predictable, compounding income structure that most healthcare businesses cannot replicate.
This post makes the full business case for the peptide therapy clinic in 2026. It covers the market data, the financial structure, the regulatory environment, what drives margin in the business, and what an entrepreneur who moves on this opportunity with the right structure behind them is actually building. Altos Consulting Group has helped launch more than 350 clinics across the United States, including peptide therapy practices in multiple markets. To see the clinics ACG has supported in this specific category, visit altosconsultinggroup.com/clinics-supported/peptide-therapy.
Why the Peptide Market Is Moving So Fast in 2026
Understanding why demand has accelerated this sharply — and this quickly — is essential context for any entrepreneur evaluating whether this opportunity is real or a passing wave. The data says it is structural. Here are the three forces converging to make 2026 a pivotal year for the peptide therapy clinic category.
The GLP-1 Normalization Effect
GLP-1 receptor agonists — semaglutide, tirzepatide, and their compounded equivalents — were the pharmaceutical story of the last three years. Thirty million Americans are now on GLP-1 medications, up from four million in 2020. The GLP-1 market hit $58 billion in 2026 and is tracking toward $201 billion by 2033. What the GLP-1 wave did that no marketing campaign could have accomplished is normalize the concept of clinic-administered peptide therapy as a legitimate, premium health intervention for the mainstream consumer.
Patients who came in for GLP-1 programs started asking about other peptides. What does BPC-157 do? What is Sermorelin? How is Ipamorelin different from what I am already taking? The GLP-1 patient population created an educated, receptive audience for the full peptide therapy category — and that audience is actively seeking clinical relationships that can serve their expanding curiosity about peptide-based health optimization.
The FDA Reclassification and the Access Window
In early 2026, HHS Secretary Robert F. Kennedy Jr. announced that approximately fourteen of the nineteen peptides placed on the FDA's Category 2 restricted list since 2023 would be returned to Category 1 — restoring legal compounding access for compounds including BPC-157, CJC-1295, Ipamorelin, Thymosin Alpha-1, and several growth hormone secretagogues that clinics had lost access to for nearly two years. The formal Federal Register notice has not yet published as of April 2026, but the policy direction is unambiguous.
This regulatory shift is creating a surge of entrepreneur interest in establishing peptide clinic infrastructure now — before the access fully opens and competition accelerates. Clinics that build their compliance framework, medical director relationships, and compounding pharmacy supplier access today are positioned to expand their formularies as the reclassification formalizes through Q2 and Q3 2026. Those who wait will be building infrastructure while their competitors are already operating with the reinstated compounds.
The Consumer Shift to Performance and Longevity
The peptide patient in 2026 is not primarily a sick patient seeking treatment. They are a performance-oriented adult — typically between 35 and 65 — investing proactively in energy levels, physical recovery, body composition, cognitive function, and what researchers increasingly call biological age optimization. This patient does not engage with the healthcare system reactively. They are seeking clinical partners who can help them stay ahead of biological decline rather than manage it after it arrives.
Seventy-three percent of global consumers now rate healthy aging as extremely or very important, according to Innova Market Insights. McKinsey's Future of Wellness research identifies proactive health investment as one of the few consumer spending categories that has demonstrated genuine resilience across economic downturns — growing through the 2008 recession and the 2020 COVID contraction when virtually every other discretionary category contracted. The peptide patient is not a trend chaser. They are a structural demographic reality.
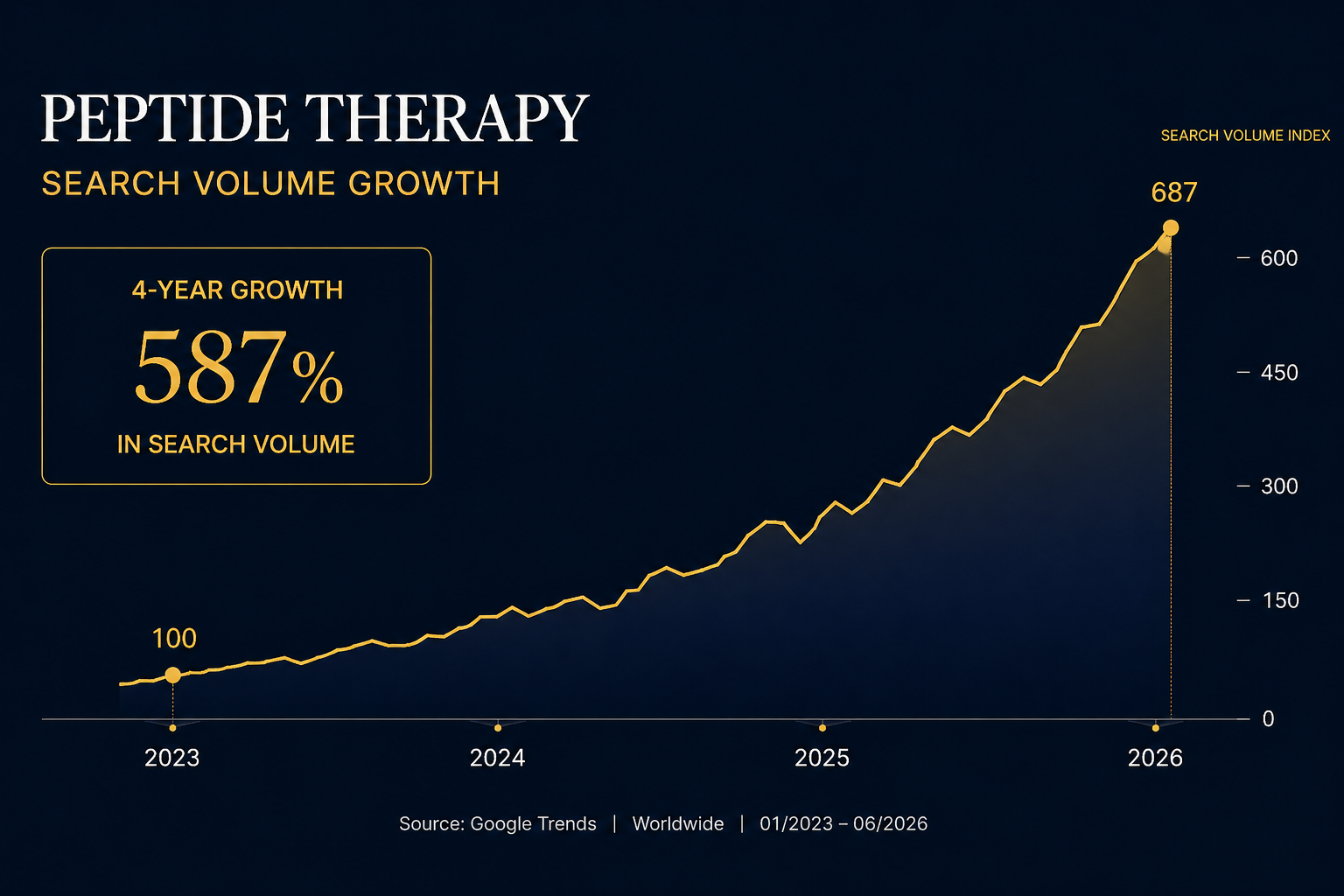
The Business Model: What Makes a Peptide Therapy Clinic Financially Durable
The financial case for the peptide therapy clinic does not rest on high patient volume. It rests on the recurring revenue structure that the protocol-based membership model produces. Understanding this distinction — between a transactional appointment business and a membership-based protocol business — is the most important strategic insight available to an entrepreneur designing a peptide clinic.
Cash-Pay Eliminates the Primary Financial Vulnerability of Traditional Healthcare
A peptide therapy clinic operates entirely in the cash-pay model. There are no insurance reimbursement cycles, no prior authorization requirements, no coding complexity, and no payer dependency. Patients pay directly for services at the point of delivery. Revenue is immediate and tied directly to patient volume — not to billing efficiency, insurance contract terms, or reimbursement timelines that can shift based on policy changes the clinic cannot control.
This structural advantage makes the peptide clinic business model fundamentally more financially predictable than insurance-dependent healthcare businesses of comparable size. When you know what you are earning on every patient interaction and that payment arrives within days rather than months, financial planning and scaling become dramatically more manageable.
Recurring Protocol Revenue vs. Transactional Appointments
The peptide clinic that structures its revenue around transactional appointments — a patient comes in, receives a service, leaves, and may or may not return — resets its revenue to zero every month. The peptide clinic that structures its revenue around protocol memberships — a patient enrolls in an ongoing program that includes compound cycles, lab monitoring, and clinical check-ins at a fixed monthly rate — builds a financial floor that compounds as the patient base grows.
A patient enrolled in a Sermorelin growth hormone optimization membership at $650 per month generates $7,800 per year in predictable recurring revenue. A patient enrolled in a comprehensive membership combining growth hormone secretagogue therapy, BPC-157 recovery protocols, and quarterly biomarker monitoring at $950 per month generates $11,400 per year. At 40 enrolled patients averaging $750 per month, the clinic generates $30,000 per month in recurring membership revenue — before additional service revenue, new patient consultations, or lab fees. That is the financial architecture of a membership-based peptide clinic operating at a modest patient volume.
These figures are illustrative planning benchmarks — actual results depend on market conditions, patient volume, pricing structure, and operational execution. They are presented to demonstrate the revenue architecture, not to project specific financial outcomes for any individual clinic.
High Gross Margins on Compounded Protocols
Compounded peptide protocols — sourced through licensed 503A and 503B compounding pharmacies under medical director prescription authority — carry gross margins that are significantly more favorable than most cash-pay health services. The margin structure is highly sensitive to sourcing relationships. Independent operators who source peptide compounds through direct retail channels pay rates that reflect their individual volume and leverage. Clinics accessing pre-negotiated supplier pricing through an established consulting partner with combined client volume can access pricing that an independent operator would typically take twelve to eighteen months of established volume history to reach.
This sourcing differential has a compounding effect on the clinic's financial model. Better per-unit compound pricing means higher gross margins at the same patient volume, which means the clinic reaches profitability faster, has more capital available for growth rather than cost recovery, and can price its membership programs competitively without sacrificing the margin structure that makes the business financially sound.

The Services That Drive the Most Revenue
A peptide therapy clinic's revenue profile is directly determined by which compounds anchor its protocol stack and how those compounds are packaged into membership programs. The following service categories represent the highest-demand, most commercially durable peptide protocol categories in 2026.
Growth Hormone Secretagogues: Sermorelin and the Ipamorelin/CJC-1295 Stack
Searches for Sermorelin increased 233 percent year-over-year as of August 2025 — making it one of the fastest-growing individual compound search categories in longevity medicine. Sermorelin works by stimulating the pituitary gland to produce and release growth hormone naturally, preserving the body's own hormonal feedback mechanisms rather than introducing exogenous growth hormone directly. Clinical benefits include improved body composition, enhanced sleep quality, accelerated recovery, and improved energy — all of which are directly relevant to the performance-oriented patient demographic that drives peptide clinic revenue.
Ipamorelin, frequently combined with CJC-1295, provides a more comprehensive growth hormone optimization protocol with a more selective mechanism and stronger IGF-1 response than Sermorelin alone. Both compounds are among those expected to be reinstated to Category 1 through the 2026 reclassification process. Growth hormone secretagogue protocols are the highest-LTV service category in the typical peptide clinic menu — patients stay enrolled for six months to multiple years and are among the strongest referral sources in the longevity patient demographic.
BPC-157: Tissue Repair and the Recovery Patient
BPC-157 — Body Protection Compound 157 — is a synthetic peptide with extensively documented preclinical evidence for tissue repair, gut integrity support, inflammatory regulation, and musculoskeletal recovery. It was placed on the Category 2 restricted list in 2023 and is expected to have legal compounding access restored through Q2 and Q3 2026.
From a business perspective, BPC-157 attracts one of the most commercially valuable patient profiles in the peptide clinic space: the active adult managing a persistent tissue injury, chronic joint issue, or gut health problem that conventional medicine has not adequately resolved. This patient is highly motivated, outcome-oriented, and willing to invest in a clinical relationship that actually moves the needle. BPC-157 protocol cycles run four to eight weeks and are frequently repeated, creating natural recurring engagement that supports the membership revenue model.
Thymosin Alpha-1: Immune Health and the Longevity Patient
Thymosin Alpha-1 has the most robust clinical track record among immune-focused longevity peptides — it holds regulatory approval in multiple countries for hepatitis and cancer adjunct therapy, providing a foundation of human trial data that most peptide compounds lack. In a clinic context, Thymosin Alpha-1 serves a patient motivated by immune resilience and longevity — a profile that does not overlap precisely with the performance and recovery patient attracted to growth hormone secretagogues and BPC-157, creating an opportunity to broaden the clinic's addressable patient demographic.
The Compliance Framework: What Every Peptide Clinic Owner Must Get Right
The single most important structural decision a peptide clinic owner makes is building the legal and compliance framework correctly before the first patient is seen. The regulatory environment for peptide therapy is actively evolving in 2026 — and the clinics that navigate this environment safely are the ones that built their operations on a proper compliance foundation rather than improvising as the rules changed around them.
The Medical Director Relationship
Every peptide compound offered to a patient at a clinical practice must be prescribed by a licensed medical professional. A medical director — typically a physician, and in states with full nurse practitioner practice authority, sometimes an NP — provides the prescribing authority and clinical oversight that makes legal peptide clinic operations possible for a non-physician owner. The medical director reviews patient labs, approves protocols, and maintains the clinical standards required for regulatory compliance. ACG facilitates medical director introductions to vetted practitioners with experience in longevity medicine and peptide therapy protocols — not just any physician willing to sign an agreement.
Compounding Pharmacy Sourcing
Every compound offered to patients must be sourced through a licensed 503A or 503B compounding pharmacy. This is not a recommendation — it is the legal baseline for operating a compliant peptide therapy practice. The gray market for research peptides — vendors selling compounds labeled for research use only — cannot be lawfully used in a clinical setting for patient care. The FDA has actively targeted gray-market peptide vendors, and clinics sourcing through these channels expose themselves, their medical directors, and their patients to significant legal and safety risk.
Documentation and Consent Standards
Patient records for peptide therapy protocols require a documented clinical rationale for each prescription, informed consent addressing the regulatory status of the compounds and the nature of the clinical relationship, baseline labs where clinically appropriate, protocol specifications, and follow-up documentation. The clinic that treats documentation as a bureaucratic inconvenience is the one that faces enforcement exposure when regulators examine the file. The clinic that treats documentation as legal and clinical protection operates sustainably across whatever regulatory changes occur.

The Market Opportunity: What the Competitive Landscape Actually Looks Like
The global peptide therapeutics market is valued at $294.58 billion in 2026 and is projected to grow at a compound annual growth rate of 8.73 percent through 2033 — nearly doubling in under a decade. The clinic-based peptide therapy segment within that broader market is the fastest-growing component, driven by the shift from retail supplements and gray-market research peptides toward clinically supervised, medically directed protocols.
What the macro market data does not capture is the local competitive picture that an entrepreneur with a specific market in mind needs to evaluate. In most mid-size and secondary U.S. markets, the number of professionally structured, compliantly operating, premium-positioned peptide therapy clinics is very small — often zero. The operators who exist tend to be independently started by practitioners who built something functional rather than something designed with business durability in mind. The market is not waiting to be created. It exists. It is simply waiting for operators who take it seriously.
Entrepreneurs who open a well-structured, correctly positioned peptide therapy clinic in 2026 are not entering a saturated category. They are establishing the standard of care in a market that does not yet have one — and that positioning, built now, compounds in value as the category grows and more consumers look for exactly what the market-leading clinic in their area already provides.
What ACG's Engagement Provides: The Difference Between Opening Correctly and Correcting Mistakes
The gap between an entrepreneur who opens a peptide therapy clinic through a structured, experienced consulting engagement and one who attempts to build it independently is not a gap in intelligence or determination. It is a gap in what they know before they make the decisions that cannot be undone. Entity structure chosen incorrectly for the state. A medical director relationship that looks functional but creates compliance exposure. Supplier pricing that makes the margin structure unworkable. A service menu built without understanding which protocols drive retention versus which ones produce one-time transactions.
Altos Consulting Group's engagement for a new peptide therapy clinic covers every layer of this decision stack. Market validation before any commitment is made. Entity structure and MSO setup through healthcare counsel appropriate to the owner's state. Medical director introduction and agreement facilitation. Supplier access at pre-negotiated pricing that reflects ACG's combined client volume rather than the leverage of a single new clinic. Clinical protocol setup and medical director sign-off. Brand and website development included in the engagement fee. Staff training covering both clinical protocols and the sales and consultation frameworks that convert patient inquiries into enrolled members. Two months of post-launch support after the doors open.
This is not a menu of optional services. It is a sequenced process designed around the specific decisions that determine whether a peptide clinic opens with durable foundations or spends its first year correcting avoidable structural problems. Everything in the engagement exists because the alternative — not having it in place — creates a specific, documented risk that ACG has seen play out in the market.
To see the peptide therapy clinics ACG has supported across the United States, visit altosconsultinggroup.com/clinics-supported/peptide-therapy. To start the conversation about your specific market and situation, visit altosconsultinggroup.com/survey.
What to Expect in the First 90 Days
The first ninety days of a peptide therapy clinic's operations are the most consequential period in its history. The patient experience standard the clinic establishes in this window — the quality of the consultations, the responsiveness of the clinical team, the organization of the protocols, the clarity of the membership onboarding process — is the standard it will spend years either reinforcing or repairing. Getting this right requires entering the opening period with systems already in place, not building them under the pressure of real patients with real expectations.
In a well-structured launch, the first thirty days are focused on consultation conversion — moving qualified leads through the consultation process into enrolled protocol memberships. Days thirty through sixty are focused on protocol delivery and early retention — ensuring that the first cohort of enrolled patients is experiencing the clinical quality that will drive referrals and long-term retention. Days sixty through ninety are focused on optimization — reviewing what is working in the consultation process, what the initial performance data shows about marketing efficiency, and what adjustments to the protocol mix or membership structure the early patient feedback suggests.
ACG's post-launch support period covers exactly this window — two months of active advisory support that includes bottleneck identification, marketing diagnosis, and protocol or operational adjustments based on real-world performance data. Not theoretical recommendations. Specific, data-driven adjustments to what the clinic is actually doing in the market it is actually operating in.
Frequently Asked Questions
Is the peptide therapy clinic market saturated in 2026?
No. Despite the significant growth in consumer search interest and patient demand, the supply of professionally operated, premium-positioned peptide therapy clinics remains significantly below market demand in most U.S. markets outside of major coastal metros. The operators entering now are establishing market-leading positions — not competing for incremental share in a crowded field.
Do I need a medical background to open a peptide therapy clinic?
No. Non-physician ownership of cash-pay peptide therapy clinics is structured in most U.S. states through the MSO model, which separates business ownership from clinical practice. The non-physician entrepreneur owns the management services organization. A licensed clinician owns the professional entity and provides prescribing authority and clinical oversight. ACG facilitates medical director introductions and ensures the entity structure is correct for the owner's state from day one.
How does the 2026 FDA peptide reclassification affect new clinic owners?
The announced reclassification of approximately fourteen Category 2 peptides back to Category 1 restores legal compounding access for compounds that many clinics had lost the ability to offer since 2023. Clinics already structured within the compounding pharmacy compliance framework — with a medical director, 503A or 503B pharmacy relationships, and proper clinical documentation — are positioned to expand their formularies as the formal rulemaking publishes through Q2 and Q3 2026. Clinics that wait to build the compliance infrastructure will be behind the access window when it fully opens.
How much does it cost to open a peptide therapy clinic?
Total startup investment for a peptide therapy clinic through ACG's structured engagement ranges from approximately $107,000 to $130,000 including the consulting fee, clinical protocol setup, technology, staffing, and initial marketing. Build-out and equipment costs vary by market and service scope. ACG partners with vetted lending providers for entrepreneurs who need financing support to reach the required startup capital.
What is the most important thing to get right before opening?
The legal structure. Entity formation, the MSO model, the Management Services Agreement between the business entity and the professional entity, and the medical director relationship must be established correctly — for the specific state the clinic will operate in — before any other commitment is made. Getting these foundational decisions wrong is the single most common and most expensive mistake a new clinic owner can make. Correcting them after the fact is significantly more time-consuming, more costly, and sometimes structurally complicated in ways that affect the clinic's ability to operate during the correction period.
Written by Nova, Senior Content Strategist at Altos Consulting Group.
